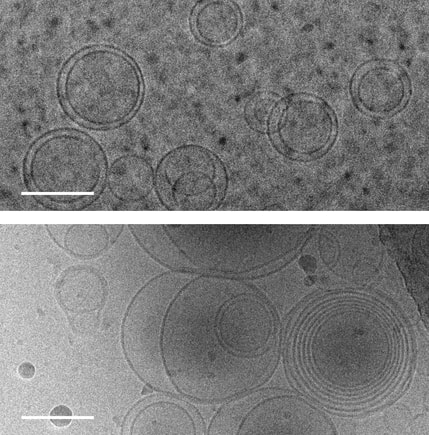
 Images of membranes (circles) taken using transmission electron cryomicroscopy. Top: membranes in a solution that contains no amino acids. Bottom: membranes in a solution containing serine, an amino acid, which triggers membranes to form multiple layers of concentric membranes. Scale bars: 100 nanometers. Images of membranes (circles) taken using transmission electron cryomicroscopy. Top: membranes in a solution that contains no amino acids. Bottom: membranes in a solution containing serine, an amino acid, which triggers membranes to form multiple layers of concentric membranes. Scale bars: 100 nanometers.
The Blue planet of ours is still the only place in universe where we know life exists! However life's journey in this planet was no less than an adventurous journey when it started 4 billion years ago when the first cells formed within a primordial soup of complex, carbon-rich chemical compounds. The primitive life forms or coacervates faced a molecular conundrum as they needed charged ions to perform their basic functions however these ions could de-stabilize and disrupt the simple membranes that encapsulated the cells.
This puzzle has been solved by a team of researchers at University of Washington and their findings were published Aug. 12 in the Proceedings of the National Academy of Sciences. The team solved this puzzle using only molecules that would have been present on the early Earth. Using cell-sized, fluid-filled compartments surrounded by membranes made of fatty acid molecules, the team discovered that amino acids, the building blocks of proteins, can stabilize membranes against magnesium ions. Their results set the stage for the first cells to encode their genetic information in RNA, a molecule related to DNA that requires magnesium for its production, while maintaining the stability of the membrane. Beside explaining the mechanism how amino acid could stabilize the membranes in unfavorable condition they went beyond and demonstrated how the individual building blocks of cellular structures — membranes, proteins and RNA — could have co-localized within watery environments on the ancient Earth. “Cells are made up of very different types of structures with totally different types of building blocks, and it has never been clear why they would come together in a functional way,” said co-corresponding author Roy Black, a UW affiliate professor of chemistry and bioengineering. “The assumption was just that — somehow — they did come together.” Prof. Black teamed up with Sarah Keller, a UW professor of chemistry and an expert on membranes. Black had been inspired by the observation that fatty acid molecules can self-assemble to form membranes, and hypothesized that these membranes could act as a favorable surface to assemble the building blocks of RNA and proteins.“You can imagine different types of molecules moving within the primordial soup as fuzzy tennis balls and hard squash balls bouncing around in a big box that is being shaken,” said Keller, who is also co-corresponding author on the paper. “If you line one surface inside the box with Velcro, then only the tennis balls will stick to that surface, and they will end up close together. Roy had the insight that local concentrations of molecules could be enhanced by a similar mechanism.” The team previously had shown that the building blocks of RNA preferentially attach to fatty acid membranes and, surprisingly, also stabilize the fragile membranes against detrimental effects of salt, a common compound on Earth past and present. The team hypothesized that amino acids might also stabilize membranes. Using variety of experimental techniques — including light microscopy, electron microscopy and spectroscopy — to test how 10 different amino acids interacted with membranes. Their experiments revealed that certain amino acids bind to membranes and stabilize them. Some amino acids even triggered large structural changes in membranes, such as forming concentric spheres of membranes — much like layers of an onion. “Amino acids were not just protecting vesicles from disruption by magnesium ions, but they also created multilayered vesicles — like nested membranes,” said lead author Caitlin Cornell, a UW doctoral student in the Department of Chemistry. The researchers also discovered that amino acids stabilized membranes through changes in concentration. Some scientists have hypothesized that the first cells may have formed within shallow basins that went through cycles of high and low concentrations of amino acids as water evaporated and as new water washed in. The new findings that amino acids protect membranes — as well as prior results showing that RNA building blocks can play a similar role — indicate that membranes may have been a site for these precursor molecules to co-localize, providing a potential mechanism to explain what brought together the ingredients for life. Keller, Black and their team will turn their attention next to how co-localized building blocks did something even more remarkable: They bound to each other to form functional machines. "That is the next step,” said Prof. Black. The study was co-authored by Gary Drobny, UW professor of chemistry; Kelly Lee, UW associate professor of medicinal chemistry; UW postdoctoral researchers Mengjun Xue and Helen Litz in the Department of Chemistry, and James Williams in the Department of Medicinal Chemistry; UW graduate students Zachary Cohen in the Department of Chemistry and Alexander Mileant in the Biological Structure, Physics and Design Graduate Program; and UW undergraduate alumni Andrew Ramsay and Moshe Gordon. The research was funded by NASA, the National Institutes of Health and the National Science Foundation.
References
1. Original article byJames Urton UW news. Note: Content may be edited for style and length. 2. Image resource: Alex Mileant/Caitlin Cornell, University of Washington.
0 Comments
Leave a Reply. |
AuthorHello! My name is Arunabha Banerjee, and I am the mind behind Biologiks. Leaning new things and teaching biology are my hobbies and passion, it is a continuous journey, and I welcome you all to join with me Archives
July 2022
Categories
All
|

 RSS Feed
RSS Feed



